Traditionally, potentiometric sensors require that two electrodes, working (WE) and reference electrode (RE), are in contact with the solution where the chemical specie to detect is present. This fact makes the sensors dependent on the selection of a very specific RE, limits miniaturization and, to work, the system requires considerable volumes of sample, as the sample must be in contact with both electrodes to close the circuit. These limitations have hindered the use of potentiometric sensors in various settings, especially in life sciences, where amperometry sensors have been widely used. Amperometry is a highly sensitive technique, but more complex than potentiometry, and in most of the applications, the sensitivity achieved by potentiometry is within the detection limits required. Therefore, the possibility of using potentiometric sensors in the detection of chemical species of biological interest, such as hydrogen peroxide, would open the door to simpler, smaller, and more economic sensors.
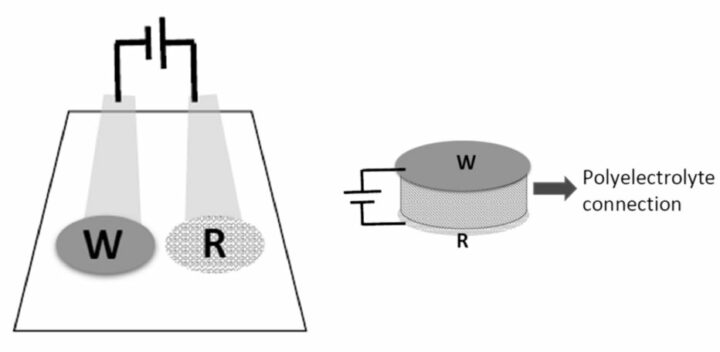
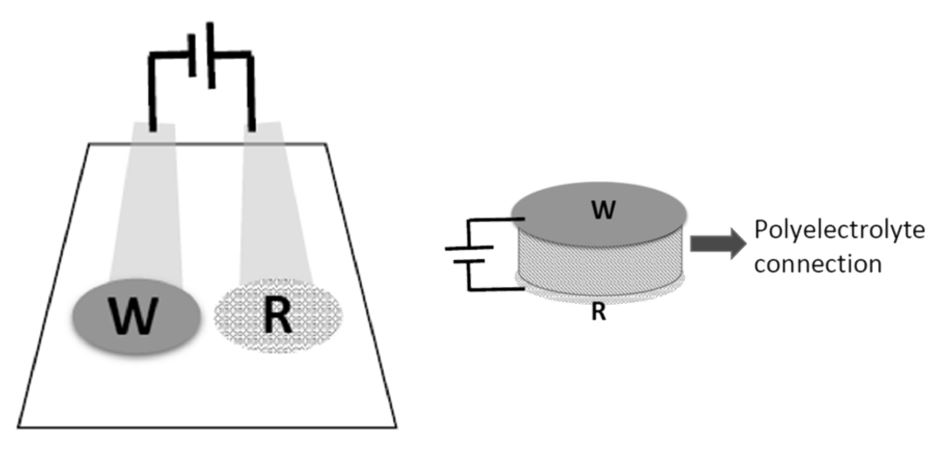
To overcome these limitations, researchers from the Laboratory of Nanosensors at the Universitat Rovira i Virgili (URV) have developed a new potentiometric sensor configuration that avoids the need of using a solution to close the circuit between the two electrodes. Instead, a polyelectrolyte bridge (made from Nafion or Aquivion) connects the WE and the RE, effectively closing the circuit without requiring a solution. This potentiometric cell can be configured in a way that the WE is positioned directly above the RE, connecting the electrodes through the polyelectrolyte bridge and avoiding the need of the RE to be in contact with the solution. The solution being only in contact with the WE, the biosensor can be miniaturized in a more compact shape.
The use of polyelectrolyte bridges allows a new configuration of more compact potentiometric sensors that provide multiple benefits:
- Use of simpler and cheaper reference electrodes.
- High miniaturization .
- Drastic reduction of volumes of sample required for measuring.
- Drastic simplification of the measures, avoiding dilution, preparation of the serum, etc.
- Cheap high scale production using Solution processing.
- More robust systems than the ones based on amperometry.
The represented institution is looking for a collaboration that leads to commercial exploitation of the presented invention.
Institution: Universitat Rovira i Virgili
TRL: 3-4
Protection status: Patent Application
Contact: Miguel Estruch / miguel@viromii.com